
Visualization of unsuccessful nuclear fusion, based on calculations by the Australian National University Thus far, reactions that created new elements were similar, with the only possible difference that several singular neutrons sometimes were released, or none at all. Two nuclei fuse into one, emitting a neutron. See also: Superheavy element § Introduction A graphic depiction of a nuclear fusion reaction. Copernicium is one of the heaviest elements whose chemical properties have been experimentally investigated. Predictions vary on whether solid copernicium would be a metal, semiconductor, or insulator. It has also been predicted to be more difficult to oxidize copernicium from its neutral state than the other group 12 elements. 
Calculations indicate that copernicium may show the oxidation state +4, while mercury shows it in only one compound of disputed existence and zinc and cadmium do not show it at all. During reactions with gold, it has been shown to be an extremely volatile element, so much so that it is possibly a gas or a volatile liquid at standard temperature and pressure.Ĭopernicium is calculated to have several properties that differ from its lighter homologues in group 12, zinc, cadmium and mercury due to relativistic effects, it may give up its 6d electrons instead of its 7s ones, and it may have more similarities to the noble gases such as radon rather than its group 12 homologues. In the periodic table of the elements, copernicium is a d-block transactinide element and a group 12 element. It was named after the astronomer Nicolaus Copernicus. Copernicium was first created in 1996 by the GSI Helmholtz Centre for Heavy Ion Research near Darmstadt, Germany. The most stable known isotope, copernicium-285, has a half-life of approximately 30 seconds. Its known isotopes are extremely radioactive, and have only been created in a laboratory. Thus, the number of neutrons in an element is obtained from the difference between the number of atomic masses and the number of atoms.283 ± 11 K ( 10 ± 11 ☌, 50 ± 20 ☏) (predicted) ģ40 ± 10 K ( 67 ± 10 ☌, 153 ± 18 ☏) (predicted)Ġ, (+1), +2, (+4), (+6) (parenthesized: prediction) Ĭopernicium is a synthetic chemical element with the symbol Cn and atomic number 112. That is, the number of atomic masses (A) = p + n Therefore, the total number of protons and neutrons is called the atomic mass number. Therefore, atomic mass refers to the total mass of protons and neutrons.Ītomic mass (A) = nucleus mass = total mass of protons and neutrons (p + n)Īgain, the mass of each proton and neutron is about 1amu. The nucleus is made up of protons and neutrons. Therefore, the mass of the nucleus is called atomic mass. One is a positively charged particle proton and the other is a charge-neutral particle neutron.Īlmost all the mass of the atom is accumulated in the nucleus. There are two types of particles in the nucleus. We already know that the nucleus is at the center of the atom. Relationship between the atomic mass and oxygen atomic number To know these properties of oxygen one must know the atomic number of oxygen. Oxygen forms bonds by receiving or sharing electrons from other elements. Therefore, oxygen is the p-block element. The last electrons of oxygen enter the p-orbital. So, the valence electrons of oxygen are six. And the last shell has a total of six electrons. The last shell of oxygen has two unpaired electrons, so the valency of oxygen is 2. The properties of an element can be determined by electron configuration. The atomic number can be used to determine the number of electrons in an element and the exact position of an element in a periodic table. Therefore, the number of negatively charged electrons orbiting in its orbit is equal to the number of positively charged protons in the nucleus.Ītomic number (Z) = Number of charges in the nucleus (p) Importance of the atomic number of oxygenĪn atomic number is a number that carries the properties of an element. That is the total number of protons in the atomic number. 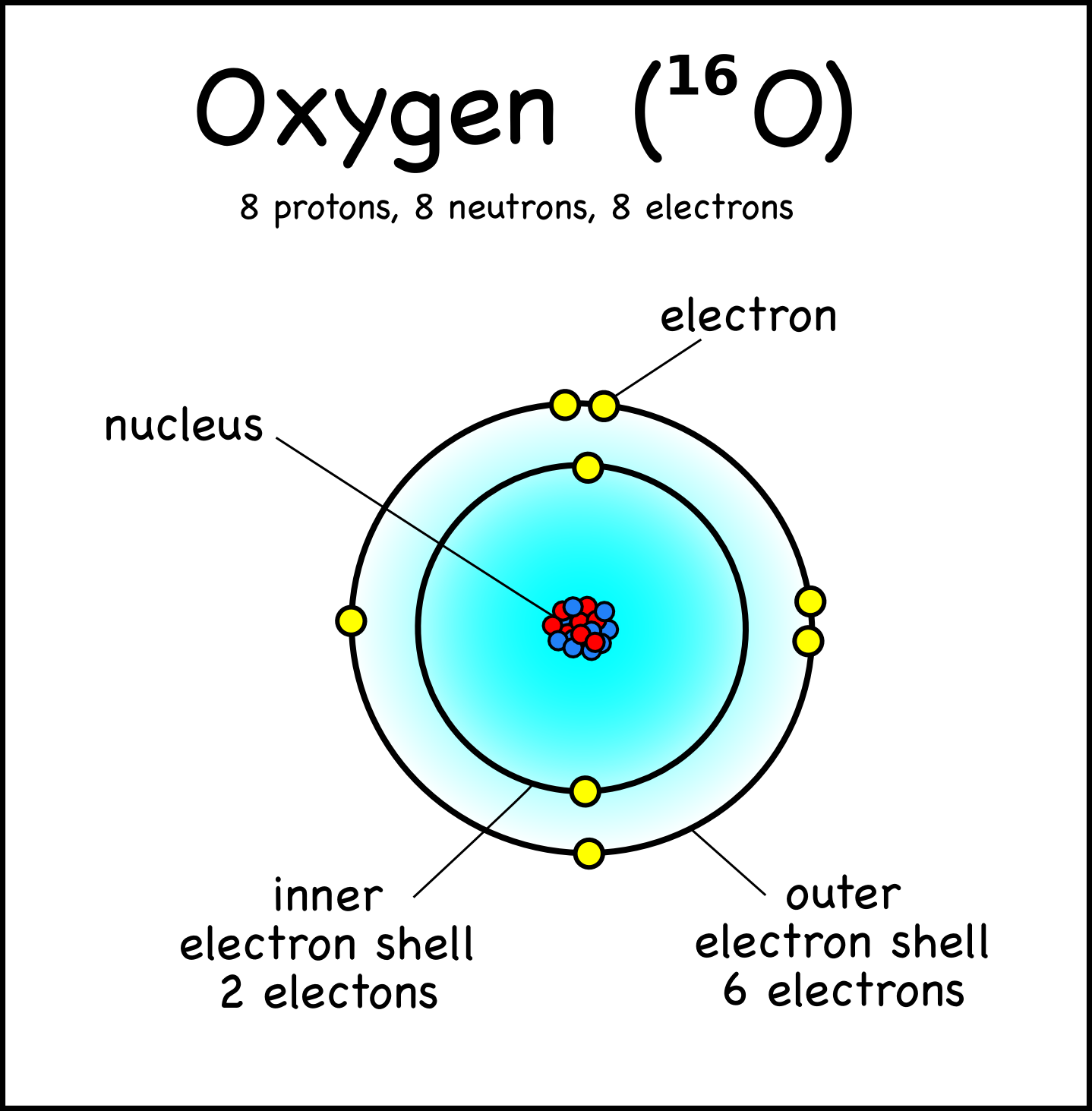
We know that protons are located in the nucleus of an atom as a positive charge. This number is equal to the serial number of the periodic table. 
The atomic number of the element is expressed by ‘Z’. Thus, the number of positive charges present in the nucleus of an element is called the atomic number of that element. He named that number the order of the atoms. The results of the experiment show that each element has a unique integer equal to the number of positive charges in the nucleus of that element. Scientist Henry Gwyn Jeffreys Moseley researched the X-ray spectrum of various elements in 1913-1914. Properties of an oxygen atom What is the atomic number? Oxygen is a p-block element and its symbol is ‘O’. Therefore, the atomic number of oxygen is 8. The oxygen atom contains a total of eight electrons and protons. The eighth element in the periodic table is oxygen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
